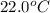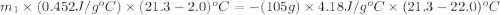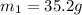Chemistry, 07.04.2020 19:02 precioushayhay
105 mL of H2O is initially at room temperature (22.0∘C). A chilled steel rod at 2.0∘C is placed in the water. If the final temperature of the system is 21.3 ∘C, what is the mass of the steel bar? Specific heat of water = 4.18 J/g⋅∘C Specific heat of steel = 0.452 J/g⋅∘C

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:10
Agas mixture with a total pressure of 745 mmhg contains each of the following gases at the indicated partial pressures: co2, 245 mmhg ; ar, 119 mmhg ; and o2, 163 mmhg . the mixture also contains helium gas. part a what is the partial pressure of the helium gas? phe p h e = nothing mmhg request answer part b what mass of helium gas is present in a 10.2-l sample of this mixture at 283 k ? m m = nothing g request answer
Answers: 1

Chemistry, 23.06.2019 05:00
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1


Chemistry, 23.06.2019 15:30
Light travels through space at 186,282 miles per second and it takes about 1.3 seconds for light to travel from the moon to earth. which of the following is the correct method of finding the distance, in miles, between the moon and earth? add 186,282 and 1.3 divide 186,282 by 1.3 multiply 186,282 by 1.3 subtract 1.3 from 186,282
Answers: 1
You know the right answer?
105 mL of H2O is initially at room temperature (22.0∘C). A chilled steel rod at 2.0∘C is placed in t...
Questions

World Languages, 18.08.2020 18:01

English, 18.08.2020 18:01

Mathematics, 18.08.2020 18:01


Social Studies, 18.08.2020 18:01


Computers and Technology, 18.08.2020 18:01



Mathematics, 18.08.2020 18:01

Biology, 18.08.2020 18:01







Mathematics, 18.08.2020 18:01


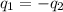
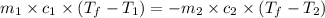
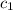 = specific heat of steel =
= specific heat of steel = 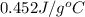
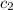 = specific heat of water =
= specific heat of water = 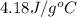
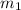 = mass of steel rod = ?
= mass of steel rod = ?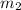 = mass of water =
= mass of water = 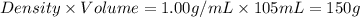
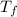 = final temperature of mixture =
= final temperature of mixture = 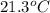
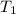 = initial temperature of steel =
= initial temperature of steel = 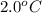
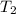 = initial temperature of water =
= initial temperature of water =