Chemistry, 07.04.2020 21:05 laura52677
G Aqueous hydrochloric acid will react with solid sodium hydroxide to produce aqueous sodium chloride and liquid water . Suppose 20. g of hydrochloric acid is mixed with 16.3 g of sodium hydroxide. Calculate the minimum mass of hydrochloric acid that could be left over by the chemical reaction. Be sure your answer has the correct number of significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
150.0 grams of asf3 were reacted with 180.0 g of ccl4 to produce ascl3 and ccl2f2. if the actual yield of ccl2f2 was 127 g, what is the percent yield?
Answers: 2

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 22.06.2019 14:30
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2
You know the right answer?
G Aqueous hydrochloric acid will react with solid sodium hydroxide to produce aqueous sodium chlorid...
Questions

Mathematics, 13.09.2020 15:01

English, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 15:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01

Geography, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

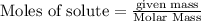
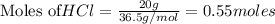
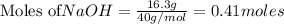
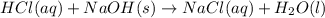
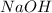 require = 1 mole of
require = 1 mole of 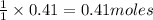 of
of 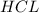 is the excess reagent.
is the excess reagent.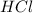 left =
left =