Chemistry, 07.04.2020 23:35 maxy7347go
How much energy must be removed from a 94.4 g sample of benzene (molar mass= 78.11 g/mol) at 322.0 K to solidify the sample and lower the temperature to 205.0 K? The following physical data may be useful. ΔHvap = 33.9 kJ/mol ΔHfus = 9.8 kJ/mol Cliq = 1.73 J/g°C Cgas = 1.06 J/g°C Csol = 1.51 J/g°C Tmelting = 279.0 K Tboiling = 353.0 K

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 10:30
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1
You know the right answer?
How much energy must be removed from a 94.4 g sample of benzene (molar mass= 78.11 g/mol) at 322.0 K...
Questions

Mathematics, 09.12.2020 19:20

Chemistry, 09.12.2020 19:20

Mathematics, 09.12.2020 19:20

History, 09.12.2020 19:20




World Languages, 09.12.2020 19:20


Social Studies, 09.12.2020 19:20


Social Studies, 09.12.2020 19:20






Geography, 09.12.2020 19:20


History, 09.12.2020 19:20

![Q=[m\times c_{p,l}\times (T_{final}-T_{initial})]+[m\times \Delta H_{fusion}]+[m\times c_{p,s}\times (T_{final}-T_{initial})]](/tpl/images/0587/9526/727a2.png)
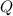 = heat released for the reaction = ?
= heat released for the reaction = ?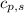 = specific heat of solid benzene =
= specific heat of solid benzene = 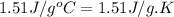
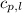 = specific heat of liquid benzene =
= specific heat of liquid benzene = 
 = enthalpy change for fusion =
= enthalpy change for fusion = 
![Q=[94.4g\times 1.73J/g.K\times (279-322)K]+[94.4g\times -125.6J/g]+[94.4g\times 1.51J/g.K\times (205-279)K]](/tpl/images/0587/9526/0dc0d.png)