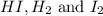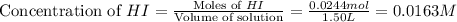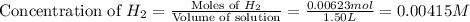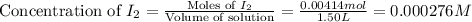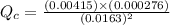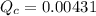Chemistry, 07.04.2020 23:09 mcckenziee
Consider the equilibrium system described by the chemical reaction below. Calculate the value of Qc for the initial set reaction conditions in a 1.50 L container: 0.00623 mol H₂, 0.00414 mol I₂, 0.0244 mol HI.\

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 22:00
8) warming your hands by a fire is an example if which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 1
You know the right answer?
Consider the equilibrium system described by the chemical reaction below. Calculate the value of Qc...
Questions


Mathematics, 26.07.2019 18:00


Biology, 26.07.2019 18:00


Biology, 26.07.2019 18:00

History, 26.07.2019 18:00

Social Studies, 26.07.2019 18:00

Chemistry, 26.07.2019 18:00











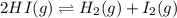
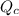 is, 0.00431
is, 0.00431![Q_c=\frac{[H_2][I_2]}{[HI]^2}](/tpl/images/0587/8224/7d2ae.png)