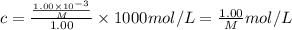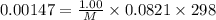A chemist is trying to determine the molar mass of a certain protein. 1.00 x 10 -3 g of it was dissolved in enough water to make 1.00 mL of solution. The osmotic pressure of this solution was found to be 1.12 torr at 25.0°C. Calculate the molar mass of the protein.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This active feature of earth's crust in building mountain ranges as well as islands. this feature is a a) cavern. b) earthquake. c) mountain. d) volcano.
Answers: 2


Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1

Chemistry, 23.06.2019 01:00
You wish to prepare a buffer consisting of acetic acid and sodium acetate with a total acetic acetate plus acetate concentration of 250 mm and a ph of 5. what concentrations of acetic acid and sodium acetate should you use
Answers: 1
You know the right answer?
A chemist is trying to determine the molar mass of a certain protein. 1.00 x 10 -3 g of it was disso...
Questions










Mathematics, 04.03.2020 04:53

Mathematics, 04.03.2020 04:53





Biology, 04.03.2020 04:54


Social Studies, 04.03.2020 04:54


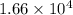 g/mol
g/mol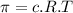
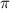 represents osmotic pressure of solution, c represents molarity of solution and T represents temperature in kelvin scale.
represents osmotic pressure of solution, c represents molarity of solution and T represents temperature in kelvin scale.