
Chemistry, 08.04.2020 19:04 vanydparis
Vinegar is a chemical used in cooking, cleaning and other common experiences. A 0.1 M solution of vinegar in water has a [H+] of about 1.3 × 10–3. (You may prefer to think of the hydronium ion concentration, [H3O+], as 1.3 × 10–3.)
A. Write the formula for the calculation of pH, and then show each step as you calculate the pH of a 0.1 M solution of vinegar.
B. Is vinegar an acid or a base? Explain how you know.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Naoki's bicycle has a mass of 10 kg. if naoki sits on her bicycle and starts pedaling with a force of 168 n, causing an acceleration of 2.8 m/s2, what is naoki's mass?
Answers: 1

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 23.06.2019 13:20
In the haber reaction, patented by german chemist fritz haber in 1908, dinitrogen gas combines with dihydrogen gas to produce gaseous ammonia. this reaction is now the first step taken to make most of the world's fertilizer. suppose a chemical engineer studying a new catalyst for the haber reaction finds that 671 liters per second of dinitrogen are consumed when the reaction is run at 271c and 0.99atm. calculate the rate at which ammonia is being produced. give your answer in kilograms per second. round your answer to significant digits.
Answers: 3
You know the right answer?
Vinegar is a chemical used in cooking, cleaning and other common experiences. A 0.1 M solution of vi...
Questions




Physics, 21.09.2020 14:01



Mathematics, 21.09.2020 14:01




History, 21.09.2020 14:01

Mathematics, 21.09.2020 14:01






Chemistry, 21.09.2020 14:01

Business, 21.09.2020 14:01

Mathematics, 21.09.2020 14:01

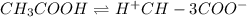
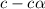
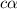
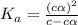
![[H^+]=c\times \alpha](/tpl/images/0589/7489/4fc41.png)
![[H^+]=0.1\times \alpha](/tpl/images/0589/7489/b5870.png)
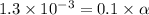
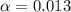
![pH=-log[H^+]](/tpl/images/0589/7489/15713.png)
![pH=-log[1.3\times 10^{-3}]=2.9](/tpl/images/0589/7489/92802.png)


