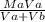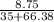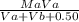Be sure to answer all parts. A 35.00−mL solution of 0.2500 M HF is titrated with a standardized 0.1318 M solution of NaOH at 25°C. (a) What is the pH of the HF solution before titrant is added? (b) How many milliliters of titrant are required to reach the equivalence point? mL (c) What is the pH at 0.50 mL before the equivalence point? (d) What is the pH at the equivalence point? (e) What is the pH at 0.50 mL after the equivalence point?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
An empty fuel tank can still contain and therefore can be even more dangerous than one full of liquid fuel.
Answers: 1

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 23.06.2019 03:30
In general metals get as you move from left to right across the periodic table.
Answers: 1

Chemistry, 23.06.2019 03:50
How many moles of potassium are needed to react completely with 12.8 moles of magnessium bromide?
Answers: 2
You know the right answer?
Be sure to answer all parts. A 35.00−mL solution of 0.2500 M HF is titrated with a standardized 0.13...
Questions

Mathematics, 12.05.2021 19:50


Biology, 12.05.2021 19:50

Mathematics, 12.05.2021 19:50



Chemistry, 12.05.2021 19:50


Mathematics, 12.05.2021 19:50

Advanced Placement (AP), 12.05.2021 19:50

History, 12.05.2021 19:50

Mathematics, 12.05.2021 19:50





English, 12.05.2021 19:50



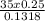
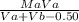
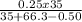
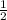 .(pKa -log [NaF])
.(pKa -log [NaF])