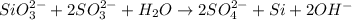Chemistry, 09.04.2020 01:50 makemybacon
SiO32- + 2SO32-+ H2O2SO42- + Si+ 2OH-In the above redox reaction, use oxidation numbers to identify the element oxidized, the element reduced, the oxidizing agent and the reducing agent. name of the element oxidized: name of the element reduced: formula of the oxidizing agent:formula of the reducing agent:

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3
You know the right answer?
SiO32- + 2SO32-+ H2O2SO42- + Si+ 2OH-In the above redox reaction, use oxidation numbers to identify...
Questions




History, 07.03.2021 05:40



History, 07.03.2021 05:40

Mathematics, 07.03.2021 05:40

History, 07.03.2021 05:40


Social Studies, 07.03.2021 05:40

Mathematics, 07.03.2021 05:40

Geography, 07.03.2021 05:40





Mathematics, 07.03.2021 05:40


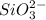 which oxidizes other but reduce itself.
which oxidizes other but reduce itself. 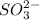 which reduces other but oxidize itself.
which reduces other but oxidize itself.