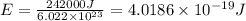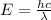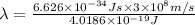Chemistry, 13.12.2019 03:31 karlamiddleschool
The energy from radiation can be used to cause the rupture of chemical bonds. a minimum energy of 242 kj/mol is required to break the chlorine–chlorine bond in cl2. determine the longest wavelength of radiation that possesses the energy to break the bond.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 22:00
8) warming your hands by a fire is an example if which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 1

Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
You know the right answer?
The energy from radiation can be used to cause the rupture of chemical bonds. a minimum energy of 24...
Questions

Computers and Technology, 17.07.2019 17:00

Computers and Technology, 17.07.2019 17:00

History, 17.07.2019 17:00

History, 17.07.2019 17:00




Mathematics, 17.07.2019 17:00


Mathematics, 17.07.2019 17:00

Chemistry, 17.07.2019 17:00

Social Studies, 17.07.2019 17:00





Computers and Technology, 17.07.2019 17:00

Computers and Technology, 17.07.2019 17:00

Geography, 17.07.2019 17:00

History, 17.07.2019 17:00

 atoms/particles/ molecules
atoms/particles/ molecules