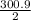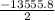Chemistry, 11.04.2020 01:50 smileyjesse6073
Calculate the enthalpy of combustion of 1 mol decane, C10H22, (l), to form CO2 and H2O. ∆Hf0 for decane is —300.9 kJ/mol using the balanced chemical equation and Appendix Table B–14.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3
You know the right answer?
Calculate the enthalpy of combustion of 1 mol decane, C10H22, (l), to form CO2 and H2O. ∆Hf0 for dec...
Questions

Social Studies, 18.11.2020 19:00


Health, 18.11.2020 19:00







Chemistry, 18.11.2020 19:00


Physics, 18.11.2020 19:00

Chemistry, 18.11.2020 19:00


Biology, 18.11.2020 19:00

Mathematics, 18.11.2020 19:00




 + 31
+ 31 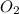 ⇒ 20 C
⇒ 20 C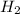 O
O