Chemistry, 14.04.2020 21:08 Qwerty2771
A chemistry student weighs out 0.154 g of chloroacetic acid (HCH2CICO2) into a 250. mL volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.1400 M NaOH solution. Calculate the volume of NaOH solution the student will need to add to reach the equivalence point.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 07:50
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 23:00
Which organism develops breathing organism develops breathing organs from pharyngeal arches? shark, spider, sea star, sea horse
Answers: 2
You know the right answer?
A chemistry student weighs out 0.154 g of chloroacetic acid (HCH2CICO2) into a 250. mL volumetric fl...
Questions

Chemistry, 18.01.2021 03:20



English, 18.01.2021 03:20

History, 18.01.2021 03:20


Mathematics, 18.01.2021 03:20

Mathematics, 18.01.2021 03:20

Mathematics, 18.01.2021 03:20


Mathematics, 18.01.2021 03:20

Mathematics, 18.01.2021 03:20

History, 18.01.2021 03:20

Health, 18.01.2021 03:20





History, 18.01.2021 03:30

Mathematics, 18.01.2021 03:30

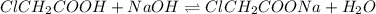
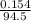 moles of chloroacetic acid
moles of chloroacetic acid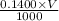 moles
moles