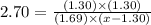For the chemical equation SO 2 ( g ) + NO 2 ( g ) − ⇀ ↽ − SO 3 ( g ) + NO ( g ) SO2(g)+NO2(g)↽−−⇀SO3(g)+NO(g) the equilibrium constant at a certain temperature is 2.70 . 2.70. At this temperature, calculate the number of moles of NO 2 ( g ) NO2(g) that must be added to 2.99 mol SO 2 ( g ) 2.99 mol SO2(g) in order to form 1.30 mol SO 3 ( g ) 1.30 mol SO3(g) at equilibrium.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 22.06.2019 21:30
How can the periodic table be used to predict the behavior of elements?
Answers: 1

Chemistry, 23.06.2019 00:30
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2

Chemistry, 23.06.2019 03:30
Ahelium balloon contains 16.9 l of helium at stp. how many atoms of helium are in the balloon
Answers: 1
You know the right answer?
For the chemical equation SO 2 ( g ) + NO 2 ( g ) − ⇀ ↽ − SO 3 ( g ) + NO ( g ) SO2(g)+NO2(g)↽−−⇀SO3...
Questions


Mathematics, 15.10.2019 10:50


Physics, 15.10.2019 10:50

French, 15.10.2019 10:50


Mathematics, 15.10.2019 10:50


Mathematics, 15.10.2019 10:50

History, 15.10.2019 10:50

Social Studies, 15.10.2019 10:50

Mathematics, 15.10.2019 10:50

Mathematics, 15.10.2019 10:50

English, 15.10.2019 10:50

Chemistry, 15.10.2019 10:50

History, 15.10.2019 10:50


History, 15.10.2019 10:50


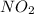 added must be, 0.37 mol
added must be, 0.37 mol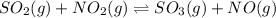
![K_c=\frac{[SO_3][NO]}{[SO_2][NO_2]}](/tpl/images/0599/9503/d13ef.png)