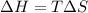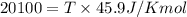Chemistry, 14.04.2020 22:27 laylac45531
For a particular reaction, Δ H ∘ = 20.1 kJ/mol and Δ S ∘ = 45.9 J / (mol ⋅ K). Assuming these values change very little with temperature,
at what temperature does the reaction change from nonspontaneous to spontaneous in the forward direction? T = K

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:20
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3


Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3
You know the right answer?
For a particular reaction, Δ H ∘ = 20.1 kJ/mol and Δ S ∘ = 45.9 J / (mol ⋅ K). Assuming these values...
Questions

Health, 16.12.2019 06:31

Chemistry, 16.12.2019 06:31

English, 16.12.2019 06:31




Mathematics, 16.12.2019 06:31


Physics, 16.12.2019 06:31







Mathematics, 16.12.2019 06:31


Mathematics, 16.12.2019 06:31


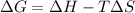
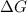 = Gibb's free energy change
= Gibb's free energy change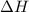 = enthalpy change = 20.1 kJ/mol = 20100 J/mol
= enthalpy change = 20.1 kJ/mol = 20100 J/mol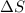 = entropy change = 45.9 J/Kmol
= entropy change = 45.9 J/Kmol