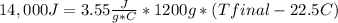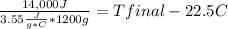Chemistry, 14.04.2020 22:50 emmaraeschool
Complete combustion of a 0.350 g sample of a compound in a bomb calorimeter releases 14.0 kJ of heat. The bomb calorimeter has a mass of 1.20 kg and a specific heat of 3.55 J/(gi°C). If the initial temperature of the calorimeter is 22.5°C, what is its final temperature? Use q equals m C subscript p Delta T..

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 23.06.2019 10:00
State the effect on the concentration of the clo- ion when there is a decrease in the concentration of the oh- ion
Answers: 1
You know the right answer?
Complete combustion of a 0.350 g sample of a compound in a bomb calorimeter releases 14.0 kJ of heat...
Questions

Physics, 01.01.2020 23:31

Biology, 01.01.2020 23:31




Chemistry, 01.01.2020 23:31


Biology, 01.01.2020 23:31

Mathematics, 01.01.2020 23:31

Mathematics, 01.01.2020 23:31


Chemistry, 01.01.2020 23:31

Biology, 01.01.2020 23:31



Mathematics, 01.01.2020 23:31


History, 01.01.2020 23:31


History, 01.01.2020 23:31

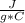 m=1.20 kg= 1200 g (1 kg=1000 g)Tfinal= ?Tinitial= 22.5 °C
m=1.20 kg= 1200 g (1 kg=1000 g)Tfinal= ?Tinitial= 22.5 °C