
Chemistry, 15.04.2020 00:47 shellxavier1
For the decomposition of phosphorous pentachloride to phosphorous trichloride and chlorine at 400K the KC is 1.1x10-2. Given that 1.0g of phosphorous pentachloride is added to a 250mL reaction flask, find the percent decomposition after the system has reached equilibrium.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1


Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3
You know the right answer?
For the decomposition of phosphorous pentachloride to phosphorous trichloride and chlorine at 400K t...
Questions










Computers and Technology, 08.02.2020 03:40



Computers and Technology, 08.02.2020 03:40







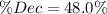
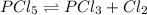
![[PCl_5]_0=\frac{1.0g*\frac{1mol}{208.24g} }{0.250L} =0.0192M](/tpl/images/0600/3535/2eefd.png)
 is introduced and the law of mass action is written as shown below:
is introduced and the law of mass action is written as shown below:![Kc=\frac{[PCl_3][Cl_2]}{[PCl_5]}=\frac{(x)(x)}{(0.0192-x)} =1.1x10^{-2}](/tpl/images/0600/3535/ffd3a.png)
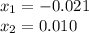
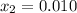 , thus, the equilibrium concentration of phosphorous pentachloride is:
, thus, the equilibrium concentration of phosphorous pentachloride is:![[PCl_5]_{eq}=0.0192M-0.01M=0.00921M](/tpl/images/0600/3535/f13fa.png)
![\% Dec=\frac{[PCl_5]_{eq}}{[PCl_5]_0}*100\% =\frac{0.00921M}{0.0192M} *100\%\\\% Dec=48.0\%](/tpl/images/0600/3535/f20dd.png)


