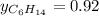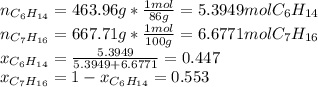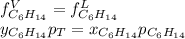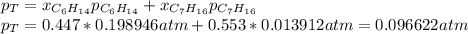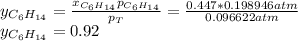Chemistry, 15.04.2020 00:51 malachilaurenc
Two linear hydrocarbons, Hexane (C6H14) and Heptane (C7H16), form pretty much an ideal solution at any composition. A solution is made at 25°C that contains 463.96 g of Hexane in 667.71 g Heptane: Characterise the vapour above this solution, and answer, What is the mole fraction of Hexane in the vapour?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Asample contains 16.75 g of the radioisotope u-236 and 50.25 g of its daughter isotope, th-232. how long did it take for decay to take place if one half-life of u-236 is 23 million years? 46 million years 69 million years 92 million years 115 million years
Answers: 3

Chemistry, 21.06.2019 21:00
Mrs. smith ordered a root beer float (vanilla ice cream + root beer). mrs. smith noticed that the three states of matter (solid, liquid, and gas) all existed simultaneously in her root beer float. a. identify each phase of matter in the root beer float. b. describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? ) c. identify one phase change you would see in a root beer float and described what causes this change.
Answers: 2

Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1
You know the right answer?
Two linear hydrocarbons, Hexane (C6H14) and Heptane (C7H16), form pretty much an ideal solution at a...
Questions




Physics, 08.08.2021 14:00


Physics, 08.08.2021 14:00

Geography, 08.08.2021 14:00


Mathematics, 08.08.2021 14:00



Mathematics, 08.08.2021 14:00

Business, 08.08.2021 14:00


Mathematics, 08.08.2021 14:00


English, 08.08.2021 14:00

Mathematics, 08.08.2021 14:00

Mathematics, 08.08.2021 14:00