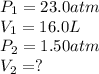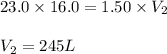Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2
You know the right answer?
A 16.0 16.0 L sample of neon gas has a pressure of 23.0 23.0 atm at a certain temperature. At the sa...
Questions


Mathematics, 27.06.2020 23:01








Mathematics, 27.06.2020 23:01










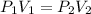
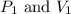 are initial pressure and volume.
are initial pressure and volume.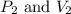 are final pressure and volume.
are final pressure and volume.