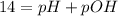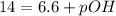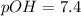Chemistry, 15.04.2020 00:57 sainijasdeep27
In pure water, some of the molecules ionize according to the equation H2O→H+ + OH−. The extent of the ionization increases with temperature. A student heats pure water and records the measured pH at 50°C as 6.6. Based on this information, what mathematical relationships gives the pOH pOH of pure water at 50°C?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3


Chemistry, 23.06.2019 00:30
What is calcium oxide+diphosphorus pentoxide--> calcium phosphate balanced
Answers: 1
You know the right answer?
In pure water, some of the molecules ionize according to the equation H2O→H+ + OH−. The extent of th...
Questions

Law, 28.02.2021 06:00

Law, 28.02.2021 06:00

History, 28.02.2021 06:00

History, 28.02.2021 06:00

Mathematics, 28.02.2021 06:00

English, 28.02.2021 06:00


Business, 28.02.2021 06:00



Mathematics, 28.02.2021 06:00

Mathematics, 28.02.2021 06:00

Mathematics, 28.02.2021 06:00

Mathematics, 28.02.2021 06:00


Advanced Placement (AP), 28.02.2021 06:00

Mathematics, 28.02.2021 06:00



Physics, 28.02.2021 06:00

 is,
is, 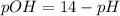
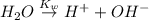
![K_w=[H^+][OH^-]](/tpl/images/0600/4070/bc68a.png)
![\log K_w=\log [H^+]+\log [OH^-]](/tpl/images/0600/4070/f0b12.png)
![-\log K_w=-\log [H^+]+(-\log [OH^-])](/tpl/images/0600/4070/00e23.png)
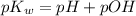
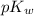 is 14 at 25-50°C.
is 14 at 25-50°C.