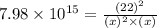Chemistry, 15.04.2020 03:10 danielburke24
The reaction SO2(g)+2H2S(g)←→3S(s)+2H2O(g) is the basis of a suggested method for removal of SO2 from power-plant stack gases. The standard free energy of each substance are ΔG∘fS(s) = 0 kJ/mol, ΔG∘fH2O(g) = -228.57 kJ/mol, ΔG∘fSO2(g) = -300.4 kJ/mol, ΔG∘fH2S(g) = -33.01 kJ/mol. If PSO2 = PH2S and the vapor pressure of water is 22 torr , calculate the equilibrium SO2 pressure in the system at 298 K.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 23.06.2019 09:20
Four statements about the development of the atomic model are shown below. a: electrons have wavelike properties. b: atoms have small, negatively charged particles. c. the center of an atom is a small, dense nucleus. d: atoms are hard, indivisible spheres. which order of statements represents the historical development of the atomic model? c-d-a-b c-d-b-a d— в-а — с d-b-c-a
Answers: 1

Chemistry, 23.06.2019 14:00
How does electronegativity changes as we move from left to right across a period
Answers: 1
You know the right answer?
The reaction SO2(g)+2H2S(g)←→3S(s)+2H2O(g) is the basis of a suggested method for removal of SO2 fro...
Questions




Mathematics, 13.07.2019 08:50


Arts, 13.07.2019 08:50

History, 13.07.2019 08:50

History, 13.07.2019 08:50


Chemistry, 13.07.2019 08:50


History, 13.07.2019 08:50


Mathematics, 13.07.2019 09:00


Mathematics, 13.07.2019 09:00

Mathematics, 13.07.2019 09:00

History, 13.07.2019 09:00

Chemistry, 13.07.2019 09:00

 pressure is,
pressure is, 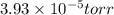
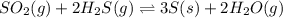
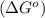 .
.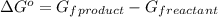
![\Delta G^o=[n_{S(s)}\times \Delta G_f^0_{(S(s))}+n_{H_2O(g)}\times \Delta G_f^0_{(H_2O(g))}]-[n_{SO_2(g)}\times \Delta G_f^0_{(SO_2(g))}+n_{H_2S(g)}\times \Delta G_f^0_{(H_2S(g))}]](/tpl/images/0600/9913/1b30c.png)
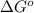 = standard free energy of reaction = ?
= standard free energy of reaction = ?![\Delta G^o=[3mole\times (0kJ/mol)+2mole\times (-228.57kJ/mol)]-[1mole\times (-300.4kJ/mol)+2mole\times (-33.01kJ/mol)]](/tpl/images/0600/9913/f2373.png)
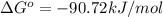
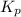
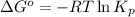
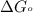 = standard Gibbs free energy = -90.72 kJ/mol
= standard Gibbs free energy = -90.72 kJ/mol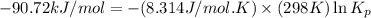
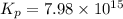
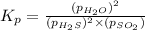
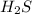 = x
= x