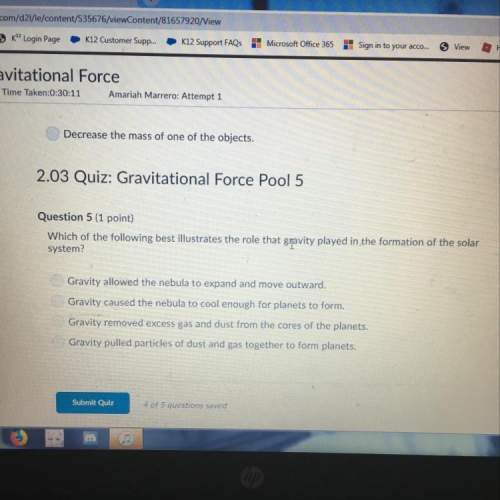
Chemistry, 15.04.2020 20:35 kps26pd2mea
Cathodic protection of iron involves using another more reactive metal as a sacrificial anode. Classify each of the following metals by whether they would or would not act as a sacrificial anode to iron.
Drag each item to the appropriate bin.
View Available Hint(s)
Reset
Help
Ag
Mg
Cu
Pb
Sn
Zn
Au
a. Will act as sacrificial anode for iron
b. Will not act as sacrificial anode for iron

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 21:00
Which property of water causes water drops to bead on a freshly waxed car?
Answers: 2
You know the right answer?
Cathodic protection of iron involves using another more reactive metal as a sacrificial anode. Class...
Questions

Mathematics, 27.12.2021 14:00

Mathematics, 27.12.2021 14:00


Biology, 27.12.2021 14:00


Mathematics, 27.12.2021 14:00

Arts, 27.12.2021 14:00


English, 27.12.2021 14:00


Mathematics, 27.12.2021 14:00

Social Studies, 27.12.2021 14:00



Physics, 27.12.2021 14:00

Arts, 27.12.2021 14:00

English, 27.12.2021 14:00

History, 27.12.2021 14:00

English, 27.12.2021 14:00