The solution would be unsaturated.
Explanation:
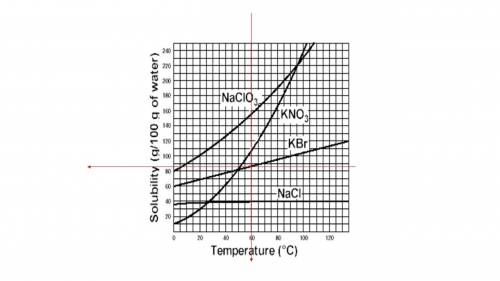
The data are garbled and difficult to decipher so i have to work with other data.
I will use the graph attached.
There, I have drawn two red lines that intersect at T = 60ºC and Solubility close the the middle point between 80 and 90 g/100g of H₂O, but a little bit above (closer to 90 than to 80 g/100g of H₂O).
That means that the solubility at 60ºC is close to 85g/100g of H₂O but a little bit above.
Then, if 85 g of KBr were dissolved in 100g of water at 60°C, the solution would be unsaturated: it can dissolve a little bit more of solute.
To understand how you use the solubility curve graphs:
For a given temperature:
move vertically upward from the mark of the temperature, i.e. 60ºC, in this case.Intersect the curve that corresponds to your solute, i.e. KBr in this casePoints on the line mean the solution is saturated.Points below the line correspond to unsaturated solutionsPoints above the line correspond to supersaturated solutions.