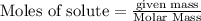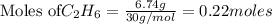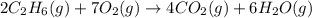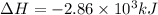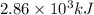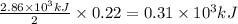Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Will give ! what are the advantages and disadvantages of nuclear power? check all that apply. one advantage of nuclear energy is that it does not produce carbon dioxide emissions. storage of nuclear waste is a short-term problem associated with nuclear energy. the problem with uranium mining is that a large quantity of uranium must be extracted to meet energy needs because the energy release from uranium fission is so low. safe operation of a nuclear power plant can be jeopardized by a human mistake.
Answers: 1

Chemistry, 22.06.2019 23:30
To find the work done, the force exerted and distance moved are multiplied. a couch is moved twice before you are happy with its placement. the same force was used to move the couch both times. if more work is done the first time it is moved, what do you know about the distance it was moved? a) when more work was done, the couch was moved the same distance. b) when more work was done, the couch was moved less. c) when more work was done, the couch was moved further. d) when more work was done, the couch wasn't moved at all.
Answers: 1

Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1
You know the right answer?
The following thermochemical equation is for the reaction of ethane(g) with oxygen(g) to form carbon...
Questions

Computers and Technology, 22.05.2020 07:02



Mathematics, 22.05.2020 07:02

Biology, 22.05.2020 07:02



Mathematics, 22.05.2020 07:02



Computers and Technology, 22.05.2020 07:02

Mathematics, 22.05.2020 07:02


English, 22.05.2020 07:02


History, 22.05.2020 07:02

Health, 22.05.2020 07:02

Mathematics, 22.05.2020 07:02

Mathematics, 22.05.2020 07:02

Mathematics, 22.05.2020 07:02

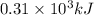 of energy is produced
of energy is produced