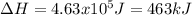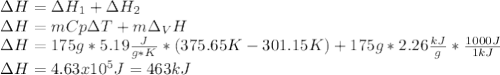Salt water can be desalinated by distillation. How much energy is needed to convert 175 g of salt water at 28.0 °C to water vapor if the specific heat of salt water is 5.19 J/g K, the boiling point of salt water is 102.5 °C, and the enthalpy of vaporization is 2.26 kJ/g?
A. 317 kJ
B. 399 kJ
C. 463 kJ
D. 512 kJ
E. 673 kJ

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 10:00
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1

Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3
You know the right answer?
Salt water can be desalinated by distillation. How much energy is needed to convert 175 g of salt wa...
Questions

Biology, 04.11.2020 22:40

Arts, 04.11.2020 22:40


Chemistry, 04.11.2020 22:40

Mathematics, 04.11.2020 22:40

Mathematics, 04.11.2020 22:40

History, 04.11.2020 22:40

Physics, 04.11.2020 22:40



Mathematics, 04.11.2020 22:40


Mathematics, 04.11.2020 22:40



Mathematics, 04.11.2020 22:40


Mathematics, 04.11.2020 22:40