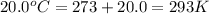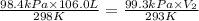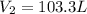Chemistry, 16.04.2020 04:43 laureneb0403
A gas balloon has a volume of 106.0 liters when the temperature is 25.0 °C and the pressure is 740.0 mm Hg. What will its volume be at 20.0 °C and 99.3kPa

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3
You know the right answer?
A gas balloon has a volume of 106.0 liters when the temperature is 25.0 °C and the pressure is 740.0...
Questions




Mathematics, 19.05.2020 10:58








Mathematics, 19.05.2020 10:58




Mathematics, 19.05.2020 10:58



Mathematics, 19.05.2020 10:58

Mathematics, 19.05.2020 10:58

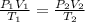
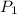 = initial pressure of gas = 740.0 mmHg = 98.4 kPa
= initial pressure of gas = 740.0 mmHg = 98.4 kPa = final pressure of gas = 99.3 kPa
= final pressure of gas = 99.3 kPa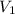 = initial volume of gas = 106.0 L
= initial volume of gas = 106.0 L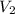 = final volume of gas = ?
= final volume of gas = ?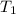 = initial temperature of gas =
= initial temperature of gas = 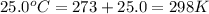
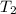 = final temperature of gas =
= final temperature of gas =