Chemistry, 16.04.2020 09:35 emmaguentherp3hjd3
A can of coke contains 25 mL of carbon dioxide gas at 100kPa. If you take it on a hike up Mount Everest and the pressure decreases to 50 kPa, what will the new volume of the carbon dioxide gas in your coke can be?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:20
What is the ima of the 1 st class lever in the graphic given? 2 3 0.5
Answers: 1


Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 23.06.2019 01:50
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1
You know the right answer?
A can of coke contains 25 mL of carbon dioxide gas at 100kPa. If you take it on a hike up Mount Ever...
Questions


Arts, 04.12.2020 17:30



Mathematics, 04.12.2020 17:30

English, 04.12.2020 17:30



History, 04.12.2020 17:30


English, 04.12.2020 17:30

Mathematics, 04.12.2020 17:30


English, 04.12.2020 17:30




Biology, 04.12.2020 17:30


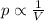
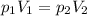
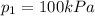 is the initial pressure of the gas in the coke
is the initial pressure of the gas in the coke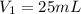 is the initial volume
is the initial volume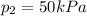 is the final pressure
is the final pressure