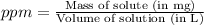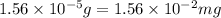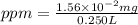Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:50
An engineering team designs a new rocket that is faster and lighter than any other model being produced. however, the materials end up being so expensive that no company can afford to buy them. which step of the engineering process should have addressed this problem? a. know the background. b. evaluate the results. c. identify a need. d. do the work.
Answers: 2

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2
You know the right answer?
A250-ml aqueous solution contains 1.56 mc025-1.jpg 10–5 g of methanol and has a density of 1.03 g/ml...
Questions

Mathematics, 05.10.2019 19:10

Social Studies, 05.10.2019 19:10





Biology, 05.10.2019 19:10


Mathematics, 05.10.2019 19:10

Mathematics, 05.10.2019 19:10


Mathematics, 05.10.2019 19:10

Chemistry, 05.10.2019 19:10

Chemistry, 05.10.2019 19:10

Mathematics, 05.10.2019 19:10


Mathematics, 05.10.2019 19:10

History, 05.10.2019 19:10

Mathematics, 05.10.2019 19:10

History, 05.10.2019 19:10