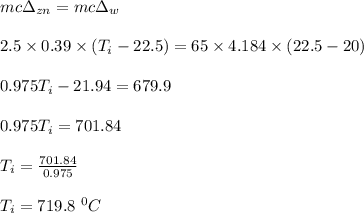A 2.50 g sample of zinc is heated, and then placed in a calorimeter containing 65.0 g of water. Temperature of water increases from 20.00 oC to 22.50 oC. The specific heat of zinc is 0.390 J/g oC. Assuming no heat loss, what was the initial temperature of the zinc metal sample?

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2
You know the right answer?
A 2.50 g sample of zinc is heated, and then placed in a calorimeter containing 65.0 g of water. Temp...
Questions


Mathematics, 12.02.2020 20:32







Advanced Placement (AP), 12.02.2020 20:32







Mathematics, 12.02.2020 20:32




Mathematics, 12.02.2020 20:32