

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

You know the right answer?
Use the following data to calculate the standard enthalpy of formation of pentane, C5H12(l)?
C...
C...
Questions

Social Studies, 09.12.2021 21:00

Mathematics, 09.12.2021 21:00

Mathematics, 09.12.2021 21:00

Health, 09.12.2021 21:00

English, 09.12.2021 21:00

Arts, 09.12.2021 21:00

History, 09.12.2021 21:00



Mathematics, 09.12.2021 21:00

Biology, 09.12.2021 21:00

English, 09.12.2021 21:00






Mathematics, 09.12.2021 21:00



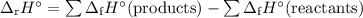
![\begin{array}{rcl}-3535.9 & = & [5\times(-393.5) -6 \times(-285.8)] - x\\-3535.9 & = & -3682.3 - x\\x& = & \textbf{-146.4 kJ/mol}\\\end{array}\\\text{The enthalpy of formation of pentane is } \large \boxed{\textbf{-146.4 kJ/mol}}](/tpl/images/0610/4927/daae3.png)


