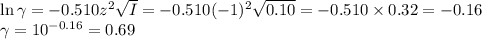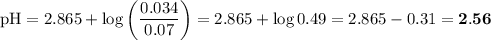We mix 0.08 moles of chloroacetic acid (ClCH2COOH) and 0.04 moles of
sodium chloroacetate (ClCH2COONa) in 1.0 L of water (pKa = 2,865).
to. Calculate the pH
yes. Calculate the pH using the formal forms (activities). Have on
counts the contribution of the protons (section a) in the calculation of the ionic strength.
C. Find the pH of a mixture prepared by dissolving the following compounds
in a final volume of 1L: 0.08 moles of ClCH2COOH, 0.04 moles of
ClCH2COONa, 0.05 moles of HNO3 and 0.06 moles of NaOH

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Write two balanced equations 1. dissolving of solid sodium hydroxide in water 2. the reaction of sodium hydroxide solution with hydrochloric acid
Answers: 1

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1
You know the right answer?
We mix 0.08 moles of chloroacetic acid (ClCH2COOH) and 0.04 moles of
sodium chloroacetate (ClC...
sodium chloroacetate (ClC...
Questions






Spanish, 10.04.2021 03:30





Health, 10.04.2021 03:30

Computers and Technology, 10.04.2021 03:30

Mathematics, 10.04.2021 03:30

Mathematics, 10.04.2021 03:30

Computers and Technology, 10.04.2021 03:30




Mathematics, 10.04.2021 03:30

Mathematics, 10.04.2021 03:30

![\begin{array}{rcl}\text{pH} & = & \text{pK}_{\text{a}} + \log \left(\dfrac{[\text{A}^{-}]}{\text{[HA]}}\right )\\\\& = & 2.865 +\log \left(\dfrac{0.04}{0.08}\right )\\\\& = & 2.865 + \log0.50 \\& = &2.865 - 0.30 \\& = & \mathbf{2.56}\\\end{array}](/tpl/images/0610/7910/ebc2c.png)
![\text{[H$^{+}$]} = 10^{-\text{pH}} \text{ mol/L} = 10^{-2.56}\text{ mol/L} = 2.73 \times 10^{-3}\text{ mol/L}](/tpl/images/0610/7910/adeed.png)
![I = \dfrac{1}{2} \sum_{i} {c_{i}z_{i}^{2}}\\\\I = \dfrac{1}{2}\left [0.04\times (+1)^{2} + 0.04\times(-1)^{2} + 0.00273\times(+1)^{2}\right]\\\\= \dfrac{1}{2} (0.04 + 0.04 + 0.00273) = \dfrac{1}{2} \times 0.08273 = 0.041](/tpl/images/0610/7910/9dfcb.png)
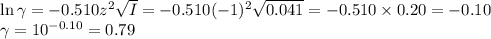
![\begin{array}{rcl}\text{pH} & = & \text{pK}_{\text{a}} + \log \left(\dfrac{a_{\text{A}^{-}}}{a_{\text{[HA]}}}\right )\\\\& = & 2.865 +\log \left(\dfrac{0.032}{0.08}\right )\\\\& = & 2.865 + \log0.40 \\& = & 2.865 -0.40\\& = & \mathbf{2.46}\\\end{array}\\](/tpl/images/0610/7910/7d363.png)
![I = \dfrac{1}{2}\left [0.10\times (+1)^{2} + 0.05 \times(-1)^{2} + 0.05\times(-1)^{2}\right]\\\\= \dfrac{1}{2} (0.10 + 0.05 + 0.05) = \dfrac{1}{2} \times 0.20 = 0.10](/tpl/images/0610/7910/85fb3.png)