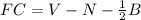Chemistry, 20.04.2020 21:15 victordhernandez01
Due to the small and highly electronegative nature of fluorine, the oxyacids of the this element are much less common and less stable than those of the other halogens. Bonding theory, however, does allow one to propose structures for these acids and use formal charges for the evaluation of these structures. For a molecule of fluorous acid, the atoms are arranged as HOFO. (Note: In this oxyacid, the placement of fluorine is an exception to the rule of putting the more electronegative atom in a terminal position.) What is the formal charge on each of the atoms?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 23.06.2019 07:00
What is the difference between covalent bonds and ionic bonds? covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the electrical attraction between charged atoms. covalent bonds involve the transfer of electrons between charged atoms; ionic bonds involve the sharing of electrons between atoms. covalent bonds involve the sharing of pairs of electrons between atoms; ionic bonds involve the sharing of single electrons between atoms. covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the sharing of protons between charged atoms.
Answers: 1
You know the right answer?
Due to the small and highly electronegative nature of fluorine, the oxyacids of the this element are...
Questions


Social Studies, 11.05.2021 21:00


Mathematics, 11.05.2021 21:00



History, 11.05.2021 21:00

Physics, 11.05.2021 21:00

Mathematics, 11.05.2021 21:00

English, 11.05.2021 21:00


Mathematics, 11.05.2021 21:00




Mathematics, 11.05.2021 21:00

English, 11.05.2021 21:00


Mathematics, 11.05.2021 21:00