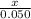Chemistry, 21.04.2020 00:04 nayelialvarez5950
Mg (s) + 2HCl (aq) → H2 (g) + MgCl2 (aq)
A:
Moles Mg: 0.050
Moles HCl: 0.050
Mass of Hydrogen gas and the limiting reactant

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 23:30
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
Mg (s) + 2HCl (aq) → H2 (g) + MgCl2 (aq)
A:
Moles Mg: 0.050
Moles HCl: 0.050
...
A:
Moles Mg: 0.050
Moles HCl: 0.050
...
Questions


English, 15.12.2020 21:50


Mathematics, 15.12.2020 21:50




Mathematics, 15.12.2020 21:50

Mathematics, 15.12.2020 21:50




Mathematics, 15.12.2020 21:50

Mathematics, 15.12.2020 21:50


Mathematics, 15.12.2020 21:50

Biology, 15.12.2020 21:50

English, 15.12.2020 21:50


Mathematics, 15.12.2020 21:50

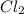 (aq)
(aq)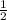 =
=