
Chemistry, 21.04.2020 00:22 donaji1024perez
1. The solubility of lead(II) chloride at some high temperature is 3.1 x 10-2 M. Find the Ksp of PbCl2 at this temperature.
2. The Ksp of Al(OH)3 is 1.0 x 10-33. What is the solubility of Al(OH)3 in 0.000010 M Al(NO3)3?
3. A precipitate of lead(II) chloride forms when 3.5 mg of NaCl is dissolved in 0.250 L of 0.12 M lead(II)nitrate.
A. True
B. False

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1

Chemistry, 22.06.2019 08:30
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1

You know the right answer?
1. The solubility of lead(II) chloride at some high temperature is 3.1 x 10-2 M. Find the Ksp of PbC...
Questions

Mathematics, 18.03.2021 19:40

Mathematics, 18.03.2021 19:40

History, 18.03.2021 19:40

Mathematics, 18.03.2021 19:40

Mathematics, 18.03.2021 19:40

Mathematics, 18.03.2021 19:40

Mathematics, 18.03.2021 19:40




Mathematics, 18.03.2021 19:40


Mathematics, 18.03.2021 19:40



Health, 18.03.2021 19:40

Mathematics, 18.03.2021 19:40



Mathematics, 18.03.2021 19:40

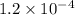 .
.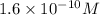 .
.
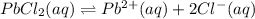
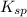
![K_{sp}=[Pb^{2+}][Cl^-]^2](/tpl/images/0613/6280/7fd11.png)
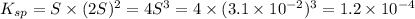
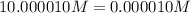
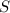
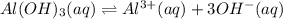
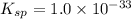
![K_{sp}=[Al^{3+}][OH^-]^3](/tpl/images/0613/6280/7ebc9.png)
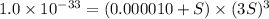
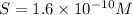
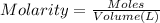
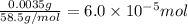
![[NaCl]=\frac{6.0\times 10^{-5} mol}{0.250 L}=0.00024 M](/tpl/images/0613/6280/43d75.png)
![[Cl^-]=[NaCl]=0.00024 M](/tpl/images/0613/6280/2673a.png)
![n=0.12 M]\times 0.250 L=0.030 mol](/tpl/images/0613/6280/4f9f1.png)
![[Pb^{2+}]=[Pb(NO_2)_3]=0.030 M](/tpl/images/0613/6280/968f2.png)
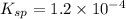
![Q_i=[Pb^{2+}][Cl^-]^2=0.030 M\times (0.00024 M)^2=1.7\times 10^{-9}](/tpl/images/0613/6280/2c9a9.png)
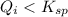 ( no precipitation)
( no precipitation)

