
Chemistry, 21.04.2020 07:00 savannahworkman11
COLLEGE CHEMISTRY 35 POINTS
Determine the percent yield when 12 g of CO2 are formed experimentally from the reaction of 0.5 moles of C8H18 reacting with excess oxygen according to the following balanced equation:
2 C8H18(l) + 25 O2(g) 16 CO2(g) + 18 H2O(l)
Please show your work.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 22.06.2019 11:00
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Chemistry, 22.06.2019 13:30
Astudent is trying to create a table that compares hypotheses, theories, and laws. hypothesis theory law do scientific researchers formulate it? yes yes yes does it explain why things happen? yes yes no yes yes yes is it used to make predictions? no yes yes which of the following questions would most likely fill the blank in the table? is it an intelligent guess? is it newly formulated? is it based on observations? has it been proved?
Answers: 1

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2
You know the right answer?
COLLEGE CHEMISTRY 35 POINTS
Determine the percent yield when 12 g of CO2 are formed experiment...
Determine the percent yield when 12 g of CO2 are formed experiment...
Questions

Mathematics, 23.04.2021 09:10

Mathematics, 23.04.2021 09:10


Mathematics, 23.04.2021 09:10









Biology, 23.04.2021 09:10



Chemistry, 23.04.2021 09:10

Mathematics, 23.04.2021 09:10

Mathematics, 23.04.2021 09:10


Mathematics, 23.04.2021 09:10

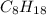 = 0.5
= 0.5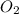 ⇒ 16 C
⇒ 16 C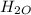
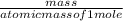
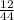
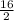 =
= 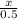
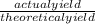 x 100
x 100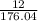 x 100
x 100

