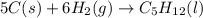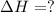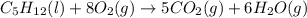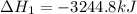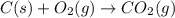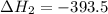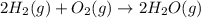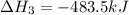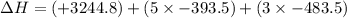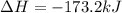Calculate Δ H rxn for the reaction: 5 C ( s ) + 6 H 2 ( g ) → C 5 H 12 ( l ) Use the following reactions and given ΔH’s: C 5 H 12 ( l ) + 8 O 2 ( g ) → 5 CO 2 ( g ) + 6 H 2 O ( g ) Δ H = − 3244.8 kJ C ( s ) + O 2 ( g ) → CO 2 ( g ) Δ H = − 393.5 kJ 2 H 2 ( g ) + O 2 ( g ) → 2 H 2 O ( g ) Δ H = − 483.5 kJ

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:50
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

You know the right answer?
Calculate Δ H rxn for the reaction: 5 C ( s ) + 6 H 2 ( g ) → C 5 H 12 ( l ) Use the following react...
Questions


Mathematics, 14.12.2021 19:00

Mathematics, 14.12.2021 19:00

Arts, 14.12.2021 19:00

English, 14.12.2021 19:00

Mathematics, 14.12.2021 19:00


Mathematics, 14.12.2021 19:00

Mathematics, 14.12.2021 19:00

Biology, 14.12.2021 19:00

History, 14.12.2021 19:00

Physics, 14.12.2021 19:00

Mathematics, 14.12.2021 19:00





Chemistry, 14.12.2021 19:00

English, 14.12.2021 19:00