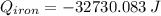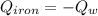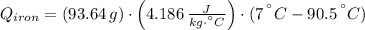Chemistry, 21.04.2020 17:37 kaylaanderson348
A piece of iron with a mass of 56.2 grams is heated and placed into a calorimeter containing 93.64 grams of water at 7.0 degrees Celsius. The final temperature of the water and the iron is 90.5 degrees Celsius. Assuming no heat is lost to the surroundings, how much heat (in Joules) does the iron release? Round your answer to the nearest 0.1 Joules.

Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
A piece of iron with a mass of 56.2 grams is heated and placed into a calorimeter containing 93.64 g...
Questions




Chemistry, 27.07.2019 22:00

Business, 27.07.2019 22:00


History, 27.07.2019 22:00





Mathematics, 27.07.2019 22:00


Chemistry, 27.07.2019 22:00

Biology, 27.07.2019 22:00

Mathematics, 27.07.2019 22:00

History, 27.07.2019 22:00