
Chemistry, 21.04.2020 17:44 LearningZone
A sample of NaOH (sodium hydroxide) contains a small amount of Na2CO3 (sodium carbonate). For titration to the phenolphthalein endpoint, 0.200 g of this sample requires 23.98 mL of 0.100 M HCl. An additional 0.700 mL of 0.100 M HCl is required to reach the methyl orange endpoint. What is the percentage of Na2CO3 by mass in the sample?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:50
Which statement describes how phase changes can be diagrammed as a substance is heated? the phase is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the phase is on the x-axis. the time is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the time is on the x-axis.
Answers: 1


Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3
You know the right answer?
A sample of NaOH (sodium hydroxide) contains a small amount of Na2CO3 (sodium carbonate). For titrat...
Questions


Mathematics, 07.05.2021 16:30

Mathematics, 07.05.2021 16:30





Geography, 07.05.2021 16:30

Mathematics, 07.05.2021 16:30

English, 07.05.2021 16:30

Mathematics, 07.05.2021 16:30


Mathematics, 07.05.2021 16:30


Mathematics, 07.05.2021 16:30

Biology, 07.05.2021 16:30

Physics, 07.05.2021 16:30


English, 07.05.2021 16:30

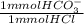 = 0.07 mmol HCO₃⁻
= 0.07 mmol HCO₃⁻


