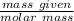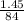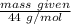Chemistry, 21.04.2020 19:26 youngchapo813p8d9u1
3. Calculate the theoretical value for the number of moles of CO2 that should have been produced in each balloon assuming that 1.45 g of NaHCO3 is present in an antacid tablet. Use stoichiometry (a mole ratio conversion must be present) to find your answers (there should be three: one answer for each balloon). (6 pts)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:10
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1

Chemistry, 23.06.2019 01:10
Volume is a measurement of how fast particles of a substance are moving
Answers: 3

Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2
You know the right answer?
3. Calculate the theoretical value for the number of moles of CO2 that should have been produced in...
Questions

History, 22.04.2021 15:50

Mathematics, 22.04.2021 15:50


Mathematics, 22.04.2021 15:50


Mathematics, 22.04.2021 15:50




History, 22.04.2021 15:50

Advanced Placement (AP), 22.04.2021 15:50





Mathematics, 22.04.2021 15:50




Mathematics, 22.04.2021 15:50

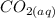 is 0.0173 moles
is 0.0173 moles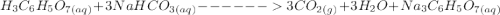
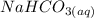 reacts with
reacts with 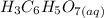 to produce 3 moles of
to produce 3 moles of