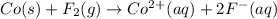Assign oxidation states to all of the species in the following redox reaction. For the reactants, identify electron loss or gain, the species oxidized, the species reduced, the oxidizing agent and the reducing agent. Co(s) + F2(g) Co2+(aq) + 2F-(aq) Oxidation state Electron loss or gain Oxidized or reduced Reducing or oxidizing agent

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3

Chemistry, 23.06.2019 02:30
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
Assign oxidation states to all of the species in the following redox reaction. For the reactants, id...
Questions

Mathematics, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30

English, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30


Social Studies, 08.04.2021 08:30


History, 08.04.2021 08:30


Computers and Technology, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30


Mathematics, 08.04.2021 08:30

History, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30

Mathematics, 08.04.2021 08:30

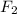 gain two electrons and thus gets reduced and acts as oxidizing agent.
gain two electrons and thus gets reduced and acts as oxidizing agent.