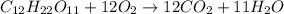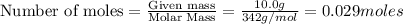Chemistry, 21.04.2020 23:31 Sillydork4545
The equation represents the combustion of sucrose. C12H22O11 + 12O2 Right arrow. 12CO2 + 11H2O If there are 10.0 g of sucrose and 8.0 g of oxygen, how many moles of sucrose are available for this reaction? 0.029 mol 0.250 mol 0.351 mol 3.00 mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
The equation represents the combustion of sucrose. C12H22O11 + 12O2 Right arrow. 12CO2 + 11H2O If th...
Questions

Biology, 26.08.2019 22:00


Biology, 26.08.2019 22:00



Mathematics, 26.08.2019 22:00


Biology, 26.08.2019 22:00

Geography, 26.08.2019 22:00





Mathematics, 26.08.2019 22:00


Social Studies, 26.08.2019 22:00




 of particles.
of particles.