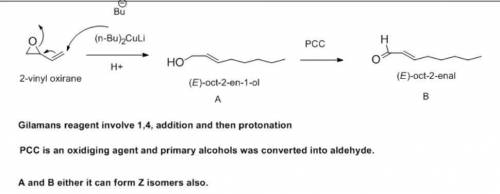
When the epoxide 2-vinyloxirane reacts with lithium dibutylcuprate, followed by protonolysis, a compound A is the major product formed. Oxidation of A with PCC yields B, a compound that gives a positive Tollens test and has an intense UV absorption around 215 nm. Treatment of B with Ag2O, followed by catalytic hydrogenation, gives octanoic acid. Identify A and B.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 09:30
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

Chemistry, 22.06.2019 12:30
The missing component to the table to the right or indicated with orange letters complete the table by filling in the corresponding numbers or symbols
Answers: 3

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1
You know the right answer?
When the epoxide 2-vinyloxirane reacts with lithium dibutylcuprate, followed by protonolysis, a comp...
Questions

Biology, 27.10.2021 03:20

Mathematics, 27.10.2021 03:20



Chemistry, 27.10.2021 03:20

Mathematics, 27.10.2021 03:20

History, 27.10.2021 03:20

Mathematics, 27.10.2021 03:20

Mathematics, 27.10.2021 03:20



Mathematics, 27.10.2021 03:20


Biology, 27.10.2021 03:30

Mathematics, 27.10.2021 03:30



English, 27.10.2021 03:30


Mathematics, 27.10.2021 03:30