
Chemistry, 22.04.2020 00:05 kailibug2287
Addition of AgNO3 to aqueous solutions of the complex results in a cloudy white precipitate, presumably AgCl. You dissolve 0.1000 g of the complex in H2O and perform a precipitation titration with 0.0500 M AgNO3 as the titrant. Using an electrode that is sensitive to [Ag ], you reach the endpoint after 9.00 mL of titrant is added. How many grams of chloride ion were present in the 0.1000-g sample

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:30
Arrange the elements in order of increasing electronegativity. use the periodic table to you arrange the elements. p o k mg
Answers: 2

Chemistry, 23.06.2019 03:00
What happens in the particles of a gas when the gas is compressed
Answers: 1

Chemistry, 23.06.2019 06:00
Complete the sentences to best explain the ranking.match the words below to the appropriate blanks in the sentences.a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1

Chemistry, 23.06.2019 19:30
What is the pressure of 5.0 mol nitrogen gas in a 2.0 l container at 268 k (the universal gas constant is 0.0821 l•atm/mol•k) a. 55 atm b. 8.8 atm c. 0.018 atm d. 220 atm
Answers: 1
You know the right answer?
Addition of AgNO3 to aqueous solutions of the complex results in a cloudy white precipitate, presuma...
Questions

Mathematics, 01.10.2019 03:30

History, 01.10.2019 03:30


History, 01.10.2019 03:30

History, 01.10.2019 03:30

Mathematics, 01.10.2019 03:30

History, 01.10.2019 03:30

History, 01.10.2019 03:30


Mathematics, 01.10.2019 03:30

Mathematics, 01.10.2019 03:30


Social Studies, 01.10.2019 03:30



Mathematics, 01.10.2019 03:30




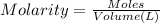
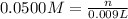
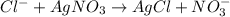
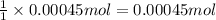 of chloride ions
of chloride ions

