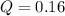Chemistry, 22.04.2020 02:39 LtotheJ0225
Nitric oxide, an important pollutant in air, is formed from the elements nitrogen and oxygen at high temperatures, such as those obtained when gasoline burns in an automobile engine. At 2000°C, K for the reaction N2(g) + O2(g) 2NO(g) is 0.01. Predict the direction in which the system will move to reach equilibrium at 2000°C if 0.4 moles of N 2, 0.1 moles of O 2, and 0.08 moles of NO are placed in a 1.0-liter container.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 23.06.2019 03:00
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
You know the right answer?
Nitric oxide, an important pollutant in air, is formed from the elements nitrogen and oxygen at high...
Questions

Mathematics, 12.02.2021 01:00

English, 12.02.2021 01:00

Physics, 12.02.2021 01:00


Mathematics, 12.02.2021 01:00



Mathematics, 12.02.2021 01:00

Mathematics, 12.02.2021 01:00


Mathematics, 12.02.2021 01:00


Mathematics, 12.02.2021 01:00



Mathematics, 12.02.2021 01:00


Mathematics, 12.02.2021 01:00

Social Studies, 12.02.2021 01:00

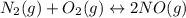
![Q=\frac{[NO]^2}{[N_2][O_2]}](/tpl/images/0617/1727/a703d.png)