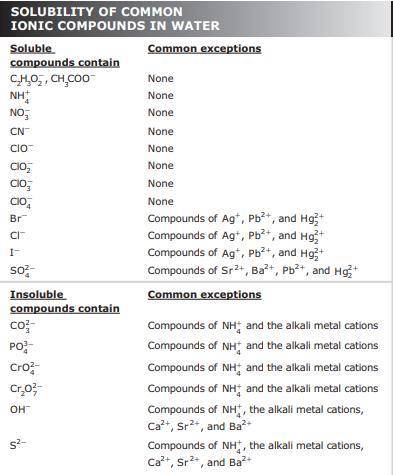
Solutions of sodium sulfate and silver nitrate are mixed together.
On a piece of scratch...

Chemistry, 22.04.2020 09:19 ConnorRecck3140
Solutions of sodium sulfate and silver nitrate are mixed together.
On a piece of scratch paper write a fully balanced double-replacement equation for this reaction.
Using the chemical equation you wrote; which answer correctly identifies the precipitate (if there is one) and the net ionic equation for the reaction that formed the precipitate?
a
Precipitate: silver sulfate
Net Ionic: 2Ag+ + SO42- --> Ag2SO4
b
There is no precipitate for the reaction; all products are soluble.
c
Precipitate: sodium nitrate
Net Ionic: Na+ + NO3- --> NaNO3
d
Precipitate: sodium nitrate
Net Ionic: Na2+ + 2NO3- --> Na(NO3)2
e
Precipitate: silver sulfate
Net Ionic: Ag+ + SO42- --> AgSO4

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2

Chemistry, 23.06.2019 06:00
Jenny wants to test the electrical conductivity of two substances dissolved in water. she is preparing the containers for the experiment. which factor is most important for her to control?
Answers: 1

Chemistry, 23.06.2019 07:00
What is the difference between covalent bonds and ionic bonds? covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the electrical attraction between charged atoms. covalent bonds involve the transfer of electrons between charged atoms; ionic bonds involve the sharing of electrons between atoms. covalent bonds involve the sharing of pairs of electrons between atoms; ionic bonds involve the sharing of single electrons between atoms. covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the sharing of protons between charged atoms.
Answers: 1
You know the right answer?
Questions


Mathematics, 12.02.2020 21:03

Mathematics, 12.02.2020 21:03




English, 12.02.2020 21:03

Physics, 12.02.2020 21:03


Computers and Technology, 12.02.2020 21:03



Mathematics, 12.02.2020 21:03






History, 12.02.2020 21:03




