
Chemistry, 22.04.2020 19:49 Xavierayala2003
When CO2(g) reacts with H2(g) to form C2H2(g) and H2O(g), 23.3 kJ of energy are absorbed for each mole of CO2(g) that reacts. Write a balanced thermochemical equation for the reaction with an energy term in kJ as part of the equation. Note that the answer box for the energy term is case sensitive. Use the SMALLEST INTEGER coefficients possible and put the energy term in the last box on the appropriate side of the equation. If a box is not needed, leave it blank. 2CO2 + 5H2 + C2H2 + 4H2O + -46.6kJ

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2


Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
You know the right answer?
When CO2(g) reacts with H2(g) to form C2H2(g) and H2O(g), 23.3 kJ of energy are absorbed for each mo...
Questions

Mathematics, 07.11.2019 02:31


History, 07.11.2019 02:31





Spanish, 07.11.2019 02:31



Geography, 07.11.2019 02:31





Mathematics, 07.11.2019 02:31

History, 07.11.2019 02:31



Mathematics, 07.11.2019 02:31

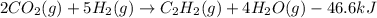
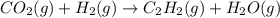
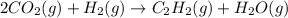
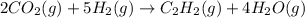
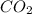 react. So, energy absorbed during the reaction is
react. So, energy absorbed during the reaction is 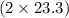 kJ or 46.6 kJ
kJ or 46.6 kJ

