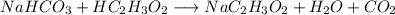Chemistry, 24.04.2020 01:54 brionesoswaldo09
You want to help your little brother make an exploding volcano for his science class. The lava will be made from reacting baking soda (NaHCO3) with vinegar (HC2H3O2). After building the volcano, you know that you want to create about 100.0g of lava (or sodium acetate, NaC2H3O2). Too little lava, and the volcano won’t overflow. Too much lava would be a giant mess! Using stoichiometry and the equation below, calculate the exact amount of baking soda needed to make 100.0g of lava. Assume you have excess vinegar. NaHCO3 + HC2H3O2 → NaC2H3O2 + H2O + CO2

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1
You know the right answer?
You want to help your little brother make an exploding volcano for his science class. The lava will...
Questions


Biology, 23.10.2020 21:30

Biology, 23.10.2020 21:30

History, 23.10.2020 21:30

History, 23.10.2020 21:30

Mathematics, 23.10.2020 21:30

Mathematics, 23.10.2020 21:30

Arts, 23.10.2020 21:30


Mathematics, 23.10.2020 21:30

German, 23.10.2020 21:30

Mathematics, 23.10.2020 21:30





Biology, 23.10.2020 21:30


English, 23.10.2020 21:30