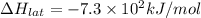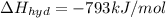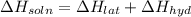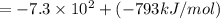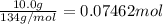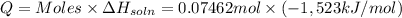Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 21:00
Which of these is an example of pseudoscience? a) predicting the time of sunrise based on data on position of earth b) predicting the date of the moon phases based on data on position of earth c) predicting eclipses based on the position of the sun and the moon d) predicting future events in a person's life based on the position of the moon
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
Lithium iodide has a lattice energy of −7.3×102kj/mol and a heat of hydration of −793kj/mol. find th...
Questions

Mathematics, 03.05.2021 21:10






Mathematics, 03.05.2021 21:10


Mathematics, 03.05.2021 21:10




Spanish, 03.05.2021 21:10

SAT, 03.05.2021 21:10

Mathematics, 03.05.2021 21:10

Mathematics, 03.05.2021 21:10

Mathematics, 03.05.2021 21:10



Mathematics, 03.05.2021 21:10