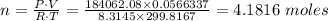Chemistry, 25.04.2020 04:05 llamawrench7366
A closed tank having a volume of is filled with 0.30 lb of a gas. A pressure gage attached to the tank reads 12 psi when the gas temperature is There is some question as to whether the gas in the tank is oxygen or helium. Which do you think it is? Explain how you arrived at your answer.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Hannah is writing a report on how albedo affects the global climate. she’s proofreading her passage for any factual errors. which sentence must hannah correct before submitting her report? earth receives energy from the sun. this energy drives many of the processes on earth, including its climate. some part of this energy is reflected by earth’s surface. we use the term albedo to describe the reflected energy. albedo of an object is the ratio of the reflected radiation to the total radiation reaching the object. a value of 0 means no energy is absorbed by the object, whereas a value of 1 means that all of the energy is absorbed. in this way, the albedo of an object can influence earth’s atmospheric temperature.
Answers: 1

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

You know the right answer?
A closed tank having a volume of is filled with 0.30 lb of a gas. A pressure gage attached to the ta...
Questions


English, 20.11.2020 19:10

Mathematics, 20.11.2020 19:10


Engineering, 20.11.2020 19:10

Advanced Placement (AP), 20.11.2020 19:10






Engineering, 20.11.2020 19:10

English, 20.11.2020 19:10

Physics, 20.11.2020 19:10


Mathematics, 20.11.2020 19:10

Mathematics, 20.11.2020 19:10

Mathematics, 20.11.2020 19:10


Mathematics, 20.11.2020 19:10