
Chemistry, 25.04.2020 23:16 kandiceboggs1
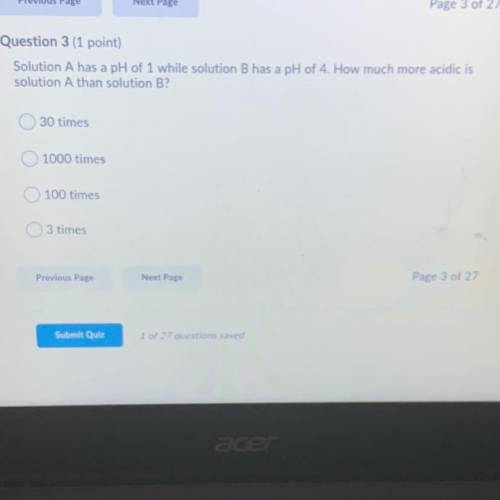
Solution A has a pH of 1 while solution B has a pH of 4.how much more acidic is solution A than solution B

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3
You know the right answer?
Solution A has a pH of 1 while solution B has a pH of 4.how much more acidic is solution A than solu...
Questions


Mathematics, 23.07.2019 17:00








History, 23.07.2019 17:00



History, 23.07.2019 17:00

Chemistry, 23.07.2019 17:00

History, 23.07.2019 17:00





Advanced Placement (AP), 23.07.2019 17:00



