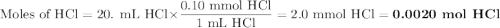Chemistry, 06.05.2020 07:17 gracelong4326
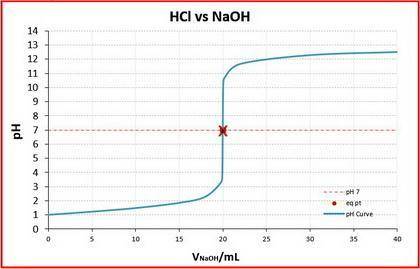
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HCl(aq) was placed in an Erlenmeyer flask. An equation for the reaction that occurs during the titration is given above.
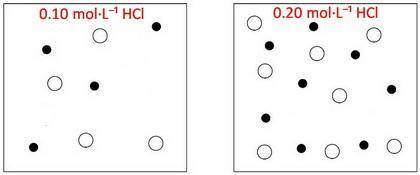
The box below to the left represents ions in a certain volume of 0.10MHCl(aq) . In the box below to the right, draw a representation of ions in the same volume of 0.20MHCl(aq)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

You know the right answer?
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HC...
Questions





Mathematics, 06.11.2021 09:10


Social Studies, 06.11.2021 09:10

French, 06.11.2021 09:10




Mathematics, 06.11.2021 09:10




History, 06.11.2021 09:20

Social Studies, 06.11.2021 09:20

Mathematics, 06.11.2021 09:20

Biology, 06.11.2021 09:20