
Chemistry, 06.05.2020 04:57 falldownguyss
If 1.332 mol of C4H10 are reacted with 6.504 mol of O2, how many mol of the excess reagent will remain unreacted? 2 C4H10 + 13 O2 ——> 8 CO2 + 10 H2O MM: 58.1 32.0 44.0 18.0

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1


Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3
You know the right answer?
If 1.332 mol of C4H10 are reacted with 6.504 mol of O2, how many mol of the excess reagent will rema...
Questions


Mathematics, 22.09.2019 11:10

History, 22.09.2019 11:10


Chemistry, 22.09.2019 11:10




Mathematics, 22.09.2019 11:10




Chemistry, 22.09.2019 11:10

Mathematics, 22.09.2019 11:10

Social Studies, 22.09.2019 11:10

Mathematics, 22.09.2019 11:10


Business, 22.09.2019 11:10

Social Studies, 22.09.2019 11:10

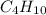 = 1.332 mol
= 1.332 mol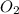 = 6.504 mol
= 6.504 mol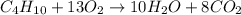
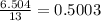 moles of
moles of 


